 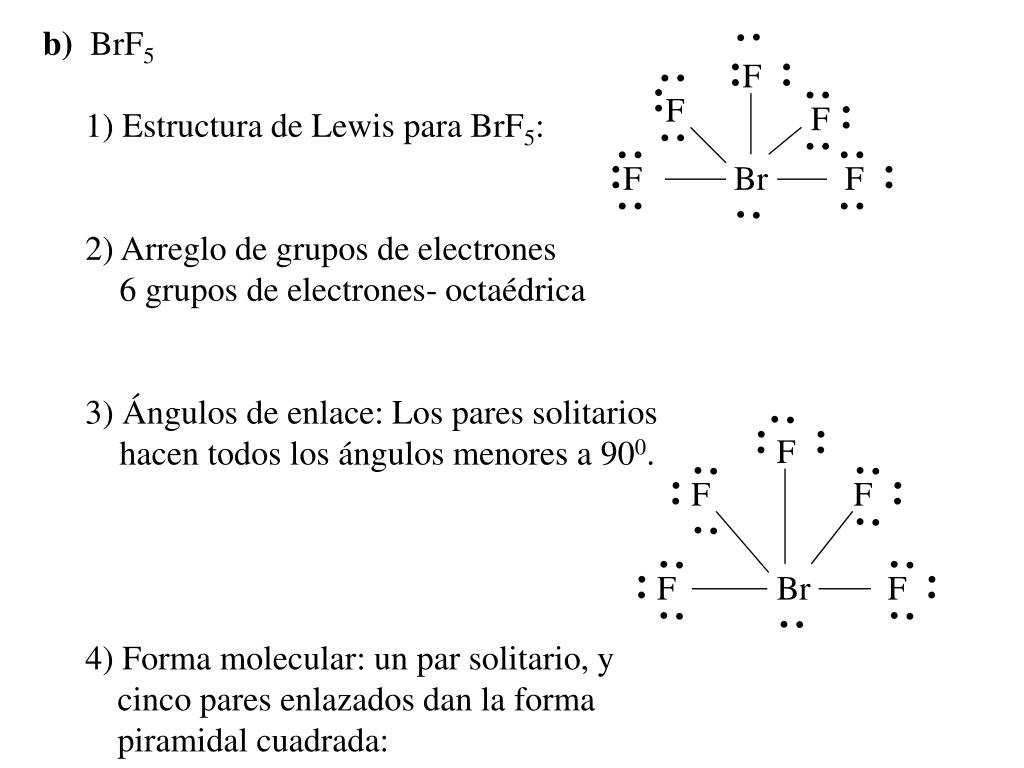
Correct The Lewis structure for XeF2 is: :-*-* b What is the electron-pair geometry for Xe.Ĭorrect The Lewis structure for XeF2 is:What is the electron-pair geometry for Xe in XeF2? What is the the shape (molecular geometry) of XeF2?What is the electron-pair geometry for N in NH2CI ? What is the the shape (molecular geometry) of NH2CI?What is the electron-pair geometry for Xe in XeF4 ? What is the the shape (molecular geometry) of XeF4 ?.Indicate the electron pair geometry and the molecular geometry for each of the six compounds listed below by completing the following table Compound Electron pair geometry Molecular geometry tetrahedral linear trigonal planar bent trigonal pyramidal Cl-S-CI: CS-CI: O: O: : C Si Cl: : Cに Indicate the electron pair geometry and the molecular geometryįor each of the six compounds listed below by completing the Indicate the electron pair geometry and the molecular geometry.What is the electron-pair geometry for Cl in CIFs There arelone pair(s) around the central atom, so the geometry of CIFs is Submit Answer Retry Entire Group 5 more group attempts remaining Previous ] There are ? lone pair(s) around the central atom, so the geometry ofAsF6 is B. Please note that "geometry" refers to the molecular or ionic geometry ?.what is the electron-pair geometry for As in Asr. Please note that "geometry" refers to the molecular or ionic geometry ?.what is the electron-pair geometry.Please note that "geometry" refers to the molecular or. What is the electron-pair geometry for Br in BrF4? There are lone pair(s) around the central atom, so the geometry of BrF4 is Use the Kelerences to access important values if needed for this question. What is the electron-pair geometry for I in IClg? There are lone pair(s) around the central atom, so the geometry of ICls is B. Please note that "geometry" refers to the molecular or ionic geometry. Please note that "geometry" refers to the molecular or ionic geometry.RESULTS (The choices of electron pair geometry and molecular shape are linear, bent, trigonal planar, trigonal pyramidal, and tetrahedral) /The lone pairs' refers to the value for the whole molecule or ion not the central atom/ valence electrons Molecule Bromine, Br bond pairs lone pairs sigma bonds pi bonds electron pair geometry molecular shape polar or nonpolar Lewis Structure: Molecule Water, H2O Lewis Structure: valence electrons bond pairs lone pairs sigma bonds pi bonds electron pair geometry molecular shape polar. RESULTS (The choices of electron pair geometry and molecular shape are linear, bent, trigonal planar, trigonal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
